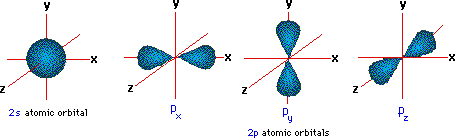
I have not yet covered that in any class, so I would not be the person to ask. As for a molecular system, I think it is best if i left that to someone else. That is an example of degeneracy in an atomic system. Thus, the n=2 energy is said to be degenerate since an electron can have this energy and be in a multitude of different eigenstates of the system. Any electron in the s orbital state or one of the p-orbital states will have this energy. Now, consider the n=2 energy for hydrogen. Thus, the n=1 energy is non degenerate since there is only one eigenstate the electron can be in (the s orbital) and have that energy. There is only one orbital(eigenstate) which has the ground energy as an eigenvalue. These additional nodes make up for the lower centrifugal potential of states with lower l as compared to states with higher l. In general ns has n-1 nodes, np n-2, nd n-3 etc. These orbitals are the eigenstates for the electron bound to the proton. To be more specific about the difference of radial functions for orbitals with different l, the 2s orbital has 1 radial node, 2p zero nodes. In the H atom there are different energy levels and within each energy level you have an orbital corresponding to different allowed values for the electron's angular momentum. A given system is said to be degenerate if more than one eigenstate has the same energy as an eigenvalue. Why are there different orientations? Because the atom is three dimensional!And … notice that the further away an orbital is from the nucleus, the more complex its shape is.The concept of degeneracy is related to the energies of the different quantum states of a system. There are seven f orbitals, all with different orientations. There are five d orbitals, four of which have a clover shape with different orientations, and one that is unique. In this Chemistry video in Hindi for class 11 we explained energies of different atomic orbitals in single-electron atom/ions as well as in multi-electron a. There are three p orbitals that differ in orientation along a three-dimensional axis. Each orbital type has a unique shape based on the energy of its electrons. Named for their energy sublevels, there are four types of orbitals: s, p, d, and f. And these sublevels consist of orbitals-specific regions of space within the sublevel where an electron is likely to be found.
DEGENERATE ATOMIC ORBITALS HOW TO
COVID-19 Portal While this global health crisis continues to evolve, it can be useful to look to past pandemics to better understand how to respond today.
Student Portal Britannica is the ultimate student resource for key school subjects like history, government, literature, and more.From tech to household and wellness products. Britannica Explains In these videos, Britannica explains a variety of topics and answers frequently asked questions.This Time in History In these videos, find out what happened this month (or any month!) in history.#WTFact Videos In #WTFact Britannica shares some of the most bizarre facts we can find.